At Seratec, quality is not just a goal—it’s the foundation of everything we do. Our approach to quality management is deeply rooted in the principles established by Joseph M. Juran in the 1990s, who pioneered the integration of quality into design to foster innovation while reducing costs associated with quality failures. Originally applied in industries such as automotive, these concepts are now central to our work in pharmaceuticals, to ensure that quality is planned and embedded throughout the entire product life cycle.
In 2003, the FDA launched an initiative, later adopted by the International Conference on Harmonisation (ICH), to bring these same principles into the pharmaceutical industry. This initiative laid the groundwork for a comprehensive approach to pharmaceutical quality management, now encapsulated in ICH guidelines Q8 through Q11. These guidelines form the backbone of our quality strategy, with the aim of integrating scientific understanding into every stage of the product lifecycle.
The ICH Q8 guideline defines «Quality by Design» (QbD) as a systematic approach to pharmaceutical development, beginning with clear objectives and emphasizing the importance of understanding the product and controlling processes through scientific principles and risk management.
The ultimate goal of regulatory authorities is to foster an efficient, agile, and flexible pharmaceutical industry, capable of producing high-quality products with minimal regulatory intervention. To achieve this, a robust quality management system (ICH Q10) is essential to support these innovative approaches.
A successful integration of QbD requires a strong quality system, as outlined in ICH Q10. At Seratec, we have long recognized the importance of such a system, embedding quality into our company culture and management strategy. We embraced the authorities’ recommendations by rethinking our design processes and investing in the ongoing training of our employees to ensure quality is embedded from the ground up.
The development of active pharmaceutical ingredients (APIs) at Seratec is guided by the principles of QbD. This involves creating detailed documentation in a master file, which requires the development of new processes that engage all aspects of the company. These processes are continuously reviewed to ensure they are both complete and effective. While we currently implement QbD in new processes, our goal is to extend this approach across all operations, supported by a range of tools and processes in line with ICH recommendations.
Guided by ICH Q9, we apply quality risk management principles to every stage of the pharmaceutical substance lifecycle, from development to distribution. This approach ensures that we provide a comprehensive understanding of both our products and their manufacturing processes, which is essential for regulatory submissions and ongoing product improvements.
Building on this, ICH Q8 (2009) recommends incorporating scientific approaches and quality risk management in pharmaceutical development, ensuring a comprehensive understanding of both the product and its manufacturing process. The knowledge gained through these practices informs regulatory submissions and allows for ongoing improvements throughout the product’s life cycle. Regulatory flexibility, particularly concerning variations, is determined by the depth of scientific understanding presented.
Finally, ICH Q11 (2012) sets out guidelines for the development and manufacturing of APIs. These scientific and technical principles help harmonize processes and ensure that the active ingredient’s quality meets regulatory standards. At Seratec, we have adopted these tools to deepen our knowledge and enhance the quality of our products.
Over the years, Seratec has developed significant scientific and technical expertise, leveraging science and risk-based decision-making to refine our approach to design and manufacturing. Our team focuses on the pharmaceutical development of APIs, considering the physicochemical and biological properties that influence drug performance. These properties, such as particle size, crystal shape, and chemical purity, are carefully designed and controlled to meet both product performance and regulatory requirements.
Pharmaceutical development of active ingredients is crucial to Seratec’s strategy. The physical, chemical, and biological properties of the API are closely linked to the drug’s performance, as defined by the Quality Target Product Profile (QTTP). These properties influence various factors, including pharmacokinetics (the fate of the drug in the body), compatibility with other components, and the inherent stability of the API. The chemical purity of the substance is a minimum standard, and impurities are often the trigger for applying the QbD approach.
By establishing a development platform for APIs, Seratec can better assess how these properties impact product performance. The resulting specifications meet both design goals and regulatory requirements.
Seratec’s commitment to quality is driven by a dedication to science and a deep understanding of the challenges we face. We are dedicated to pushing the boundaries of scientific research and development while ensuring regulatory compliance. Our research and development efforts are aligned with regulatory expectations, engaging the full expertise of our team to achieve our short-term goals and drive our business forward. By following a strategy of excellence, we ensure that quality remains at the core of everything we do.

Seratec specializes in areas ranging from developing active molecules to offering turnkey solutions for APIs.
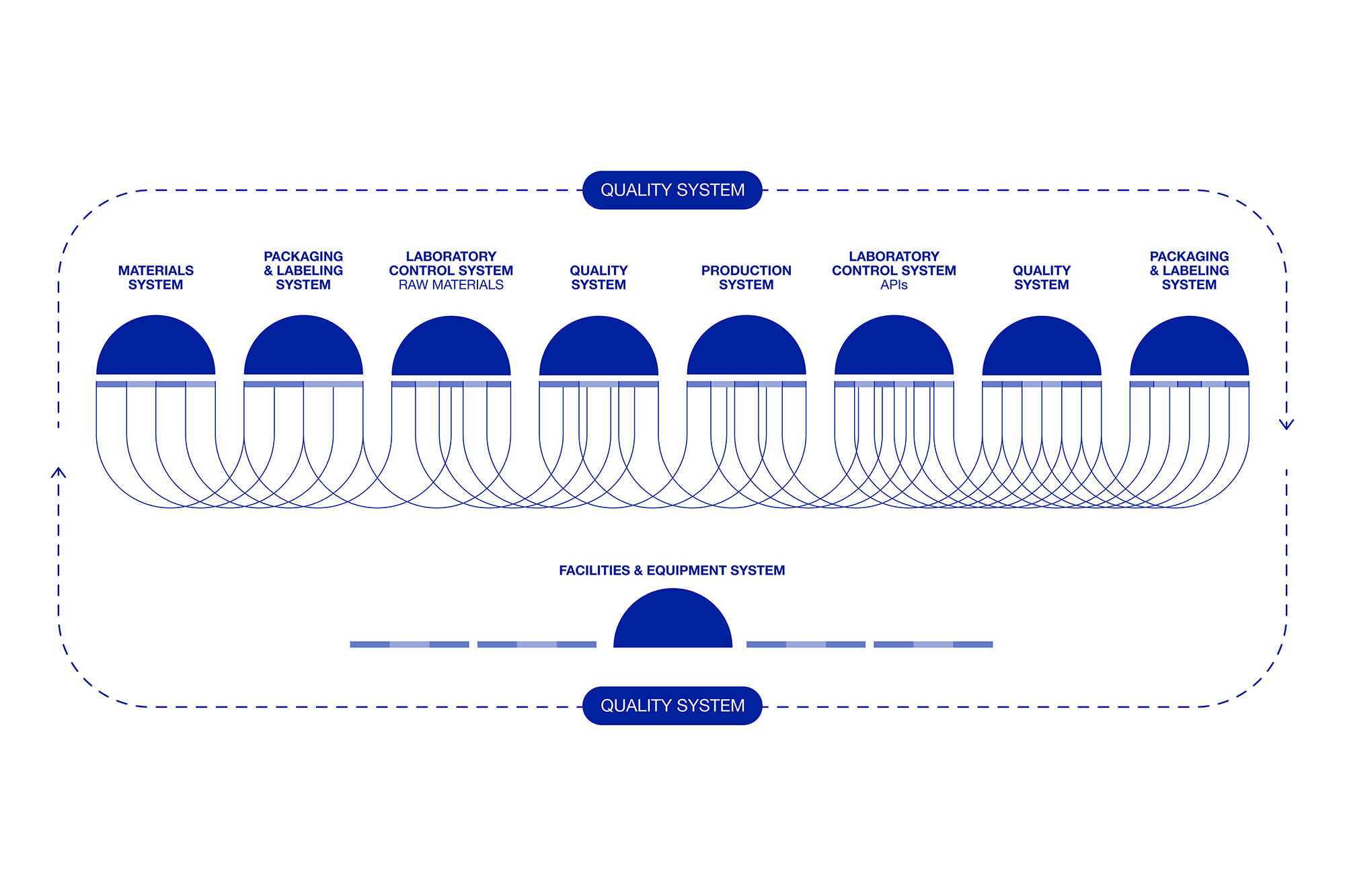
The FDA ensures all pharmaceutical products, including APIs, meet top quality and safety standards.
An active pharmaceutical ingredient (API) is a substance or mixture used in medicine production, acting as the component responsible for therapeutic effects.